
Glaucoma
Glaucoma refers to a family of eye disorders that cause damage to the optic nerve – an essential
part of the eye responsible for transmitting visual information to the brain.1,2 The damage caused
by glaucoma is irreversible and is most commonly due to abnormally high intraocular pressure
(IOP).1,3,4 The disease is an incurable cause of vision loss that exists as two major types: open- or
closed-angle glaucoma based on how it affects the anatomy of the anterior chamber of the eye.1,2
About Primary Open-Angle Glaucoma and Ocular Hypertension
The most common type is primary open-angle glaucoma (POAG) which occurs when the eye cannot properly drain fluid resulting in an increase in the IOP.1,2 Glaucoma is the leading cause of blindness worldwide.3 An estimated 52.7 million people are affected by POAG worldwide and cases of POAG are likely to increase to 79.8 million by 2040 due to the aging population.3 Currently, an estimated three million Americans are living with glaucoma, of whom 2.2 million are affected by POAG.5-7

An estimated 52.7 million people are affected by primary open-angle glaucoma (POAG) worldwide.3


Unfortunately, POAG may be asymptomatic until a relatively late stage and diagnosis is often delayed.2 When undiagnosed and untreated, POAG can gradually lead to severe visual impairment and blindness.8 One study (N=592) showed the cumulative incidences of blindness in at least one eye and bilateral blindness from glaucoma were 26.5% and 5.5% after 10 years and 38.1% and 13.5% after 20 years, respectively.9 In another study (N=122), limitations on activity due to moderate and severe glaucoma had a negative impact on patient quality of life.10
Ocular hypertension is defined as a IOP over 21 mmHg without the damage of the optic disk and view field but it is most likely to develop into open-angle glaucoma.4
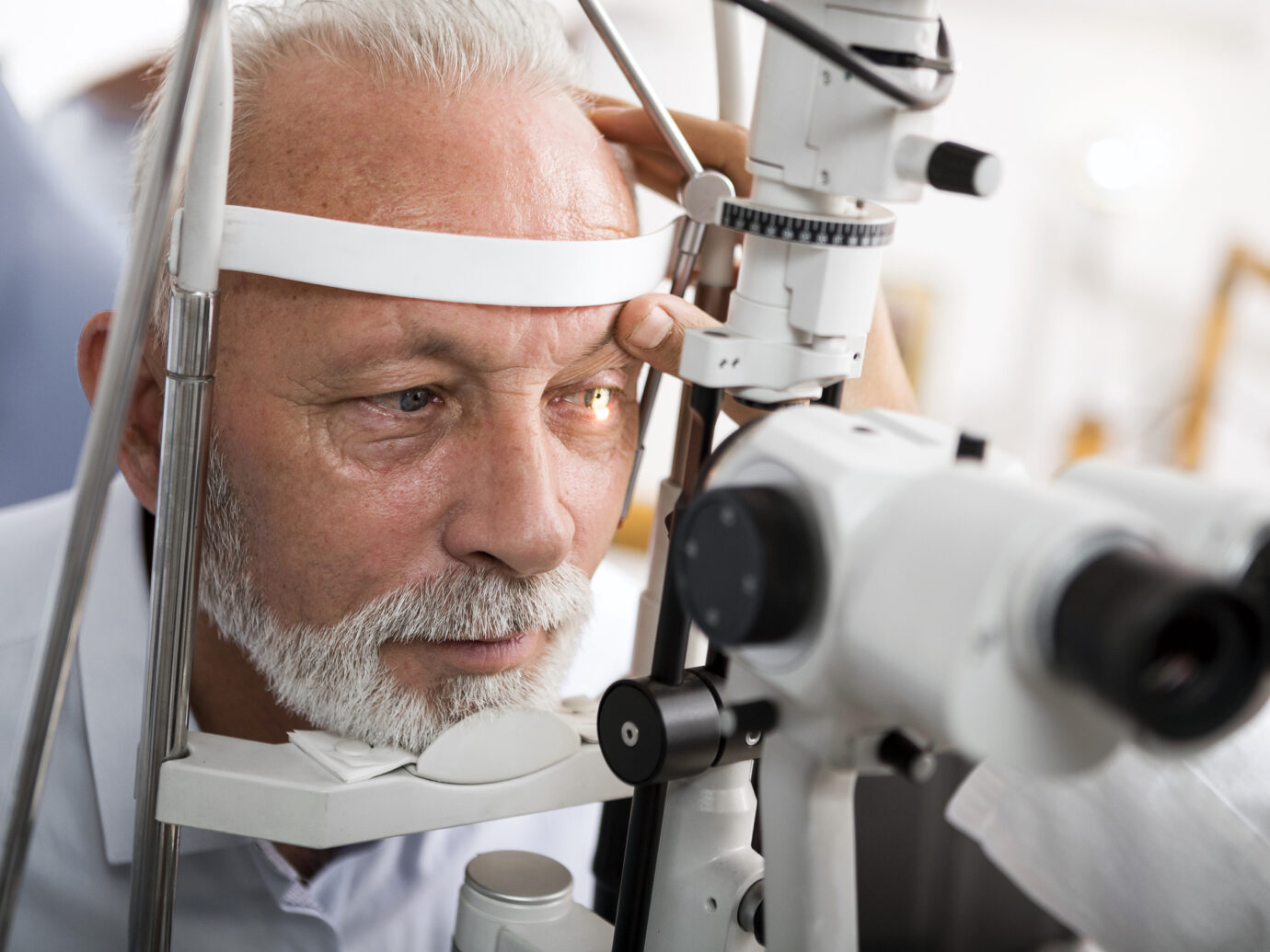
Existing Treatments
Studies have shown that IOP-lowering treatments help reduce the risk of developing POAG and may slow the progression of the disease.11 Topical therapies may include prostaglandin analogs, rho kinase inhibitors, β-adrenergic blockers, α-2 adrenergic agonists, and carbonic anhydrase inhibitors.4 Unfortunately, many patients do not achieve the potential therapeutic benefit of topical therapies due to the high rate of noncompliance.12

Challenges with Glaucoma Therapy
Existing topical glaucoma treatments require life-long daily administration which creates a high treatment burden and often leads to high rates of noncompliance, disease progression and vision loss.13-17 Among glaucoma patients, noncompliance rates range from 30-80% and have been shown to worsen with time.13,16-18 Reported barriers to compliance include difficulty with self administration, cost, forgetfulness, fear or denial, lack of understanding about glaucoma, and regimen complexity.12,19 Worsening of POAG is associated with at least 2-fold higher annual eye-related outpatient costs and significantly higher odds of falls or fractures when compared to ocular hypertension patients representing a sizable economic burden in indirect medical costs.20,21 Other concerns with topical glaucoma medications are damage to the ocular surface by active pharmaceutical agents and preservatives.22,23 Improving patient compliance with topical glaucoma medication may result in slower deterioration in visual function over time.24
REFERENCES: 1. National Eye Institute. Types of Glaucoma. Updated September 10, 2021. Accessed November 15, 2022. https://www.nei.nih.gov/learn-about-eye-health/eye-conditions-and-diseases/glaucoma/types-glaucoma. 2. Weinreb RN, et al. JAMA. 2014;311:1901-11. 3. Tham YC, et al. Ophthalmology. 2014;121(11):2081-2090. 4. Wang T, et al. Front Pharmacol. 2021;12:749858. 5. Friedman DS, et al. Arch Ophthalmol. 2004;122(4):532-538. 6. Gupta P, et al. Invest Ophthalmol Vis Sci. 2016;57(6):2905–2913. 7. US Census Bureau. Table 1. Population by Age and Sex: 2020. Washington, D.C.: U.S. Census Bureau; 2020. 8. Susanna R Jr, et al. Transl Vis Sci Technol. 2015;4:1. 9. Peters D, et al. Am J Ophthalmol. 2013;156:724-30. 10. Nayyar S, et al. Indian J Ophthalmol. 2022;70:552-557. 11. Qureshi R, et al. Ophthalmol Glaucoma. 2021;4:454-462. 12. Zaharia AC, et al. J Pers Med. 2022;12(4):514. 13. Nordstrom BL, et al. Am J Ophthalmol. 2005;140(4):598-606. 14. Schwartz GF, et al. Surv Ophthalmol. 2008;53 Suppl1:S57-68. 15. Kaur D, et al. J Curr Glaucoma Pract. 2012;6:9-12. 16. Sleath B, et al. Ophthalmology. 2011;118(12):2398-2402. doi:10.1016/j.ophtha.2011.05.013 17. Rossi GCM, et al. Eur J Ophthalmol. 2011;21(4):410-414. doi:10.5301/EJO.2010.6112 18. Olthoff CMG, et al. Ophthalmology. 2005;112(6):953-961.e7. doi:10.1016/j.ophtha.2004.12.035 19. Winfield AJ, et al. Br J Ophthalmol. 1990;74:477-80. 20. Shih V, et al. Ophthalmol Glaucoma. 2021;4:490-503. 21. Poulsen PB, et al. International Congress Series. 2005;1282:262–266. 22. Fineide F, et al. Ocul Surf. 2022;26:19-49. 23. Harasymowycz P, et al. Adv Ther. 2021;38:3019-3031. 24. Shu YH, et al. J Glaucoma. 2021;30:1047-1055.