Clinical Trials
Ocular's team is aiming to redefine drug development

REDEFINING DEVELOPMENT
Our complementary registrational trials to advance wet age-related macular degeneration (AMD) and diabetic retinopathy (DR) treatment

AXPAXLI’s potential in wet AMD:The SOL registrational trials are designed to showcase durability, repeatability, and flexibility
The SOL-X open-label extension trial evaluates long-term safety and potential disease-modifying impact
 | AXPAXLI’s potential in wet AMD: The SOL registrational trials are designed to showcase durability, repeatability, and flexibility The SOL-X open-label extension trial evaluates long-term safety and potential disease-modifying impact |

Superiority study comparing a single AXPAXLI™ (also known as OTX-TKI), an investigational axitinib hydrogel administered by intravitreal injection, dose to a single aflibercept (2mg) dose at Week 36
Design:
Two-arm trial with ~300 total subjects randomized 1:1
Primary Endpoint (Week 36):
Demonstrate that a single AXPAXLI dose is superior to a single aflibercept 2mg dose based on proportion of subjects who maintained visual acuity, defined as <15 ETDRS (Early Treatment Diabetic Retinopathy Study) letters of BCVA (best-corrected visual acuity) loss from baseline


Non-inferiority study comparing AXPAXLI Q24W to aflibercept (2mg) Q8W at Week 56
Design:
Three-arm trial with 555 total subjects randomized 2:2:1
Primary Endpoint (Week 56):
Demonstrate that a single AXPAXLI dose is non-inferior to fixed-dose aflibercept 2mg Q8W with respect to mean change from baseline in BCVA (Best-Corrected Visual Acuity) Week 56
Non-inferiority margin for the lower bound is -4.5 letters in BCVA at Week 56
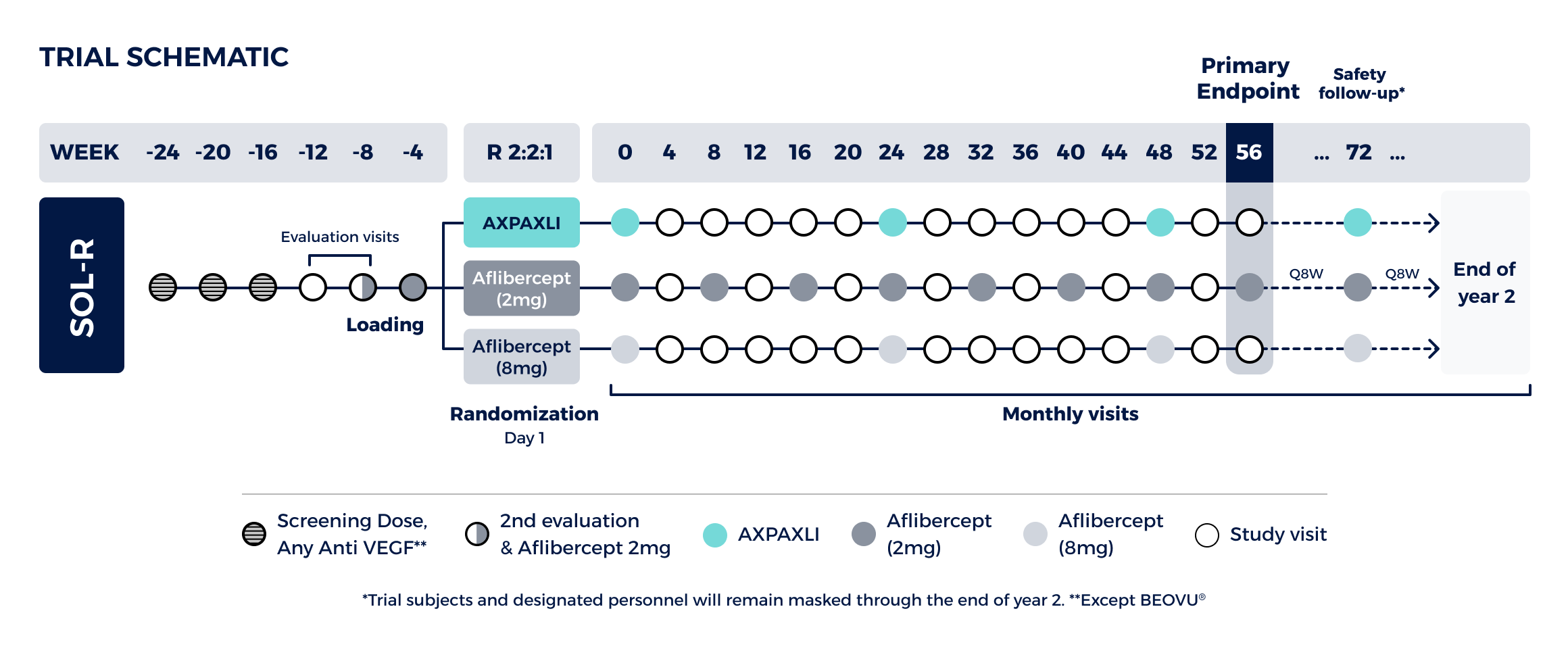

Design:
Three-year follow-up with AXPAXLI Q24W to assess long-term outcomes
Key Objectives:
Demonstrate reduction in fibrosis associated with chronic exudation
Explore long-term maintenance of visual acuity
Investigate effects of delaying AXPAXLI treatment


AXPAXLI’s potential in DR:The complementary HELIOS registrational trials leverage a novel ordinal endpoint and are designed to unlock the diabetic market and support a broad DR label
 | AXPAXLI’s potential in DR: The complementary HELIOS registrational trials leverage a novel ordinal endpoint and are designed to unlock the diabetic market and support a broad DR label |

Superiority study comparing a single AXPAXLI, an investigational axitinib hydrogel administered by intravitreal injection, compared to a single ranibizumab (0.3 mg) injection at Week 52
Design:
Study of AXPAXLI in ~432 subjects with moderate to severe non-proliferative diabetic retinopathy (NPDR) without center-involved (CI) diabetic macular edema (DME) randomized 1:1
Primary Endpoint (Week 52):
Ordinal DRSS 2-step change status at Week 52 from baseline (≥ 2 step improvement, ≥ 2-step worsening, less than 2-step change in either direction)


Design:
Study of AXPAXLI in ~930 subjects with moderate to severe NPDR without CI-DME randomized 1:1:1
Primary Endpoint (Week 56):
Ordinal DRSS 2-step change status at Week 52 from baseline (≥ 2 step improvement, ≥ 2-step worsening, less than 2-step change in either direction)

Nothing contained herein should be considered a solicitation, promotion, or advertisement for any drug including the ones under development on this website.
All investigational product candidates are currently undergoing clinical evaluation. This content is not intended to convey any conclusion of safety or efficacy, and there is no guarantee that any product candidate will successfully complete development or gain FDA approval or other regulatory authority approval.
